Parsagen ROM Test - Rupture of Membranes Test (Self Test At Home, OTC)
for solution with excellence
USD29.9
free international shipping
*We reserve right to refund order if order fails to pass internal quality control. **Regulated by the California HCQA Act ***Paypal is not stable for medical product sales. We use back up accounts from different countries. No Need for Repeated Try if payment fails. Please wait and we will fix it.

Get Coupon Now

The Parsagen ROM test is an amniotic fluid test used to aid in the detection of ROM in pregnant women reporting signs, symptoms, or complaints suggestive of ROM.
Patients with a positive Amnisure test had significantly higher rates of adverse pregnancy and neonatal outcomes (e.g., impending preterm delivery, intra-amniotic infection/inflammation, and neonatal morbidity) than those with a negative test.


The Parsagen Diagnostics ROM Test uses the principle of immunochromatography to detect the highly specific protein present in amniotic fluid. The test uses highly sensitive monoclonal antibodies that detect even a minute amount of detection target, which is present in cervicovaginal secretions after rupture of fetal membranes.
Produced by the decidua and secreted in the amniotic fluid, the specific test target was chosen as a biochemical marker for ruptured sacs due to its unique characteristics:
-High concentration in the amniotic fluid;
-Low concentration in the blood;
-Extremely low basal concentration in cervicovaginal secretions when fetal membranes are intact.
Parsagen Diagnostics ROM Test® is specific, simple and convenient
-Only diagnosis for detection target protein that correlates 99% with intra-amniotic injection of indigo carmine, recognized by ACOG as the gold standard test for PMR
-Sensitive (99%) and specific (98%), to support an accurate diagnosis with positive results and RPM negatives.
-Consistent performance throughout the gestational period
-Saves time and costs related to other tests for PRM
-There is no need for an additional confirmatory test for a positive ROM Test result.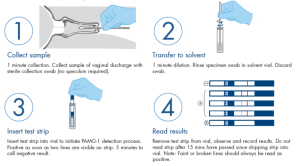
Kit comprising:
– 1 ROMTest strip in aluminum packaging
– 1 sterile vaginal swab in ETO, packaged in surgical grade paper
– 1 plastic test tube with solvent solution containing: 0.9% NaCl, 0.01% Triton X -100, 0.05% NaN3.
Note: The above information is for reference use only. Please refer to the product insert provided with the products before use.











